|
Therefore, the significance of this process is crystal clear. 
However, it gets mixed with other gases in the atmosphere, which makes it suitable for the breathing process. For human beings, it is difficult to breathe pure oxygen. Diluting them doesn’t affect their effectiveness in cleaning dishes or cookware.ĭilution is a commonly followed practice for diluting any solution, gas, or vapor. However, it is used in its concentrated form in rocket propellants.ĭishwashing liquids need dilution as they are too concentrated. It is sold at pharmacies to be used as a disinfectant or a bleaching agent. What is the molarity of the resulting solution?ĭata Given: C initial = 5ppm, V initial = 8mL, V final = 10mL, C final = ?Ĭ initial × V initial = C final × V finalĭiluted hydrogen peroxide (H2O2) is stored in concentrated form in laboratories, because its diluted form is voluminous. 
You take 8mL of water from this vessel and add 2mL of distilled water to it. An example of a dilution calculation using the Tocris dilution calculator Enter 10 into the Concentration (start) box and select the correct unit (millimolar). V1 volume to be removed from the concentrated diluted solution. 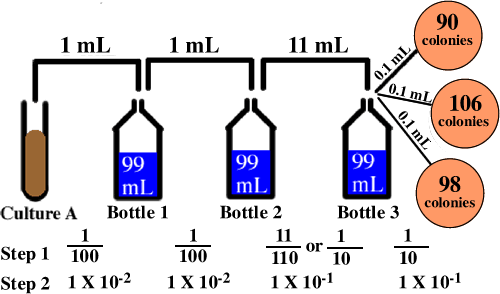 
Pulverized iron is added to a vessel filled with water. C1 concentration (molarity) of the diluted solution.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
